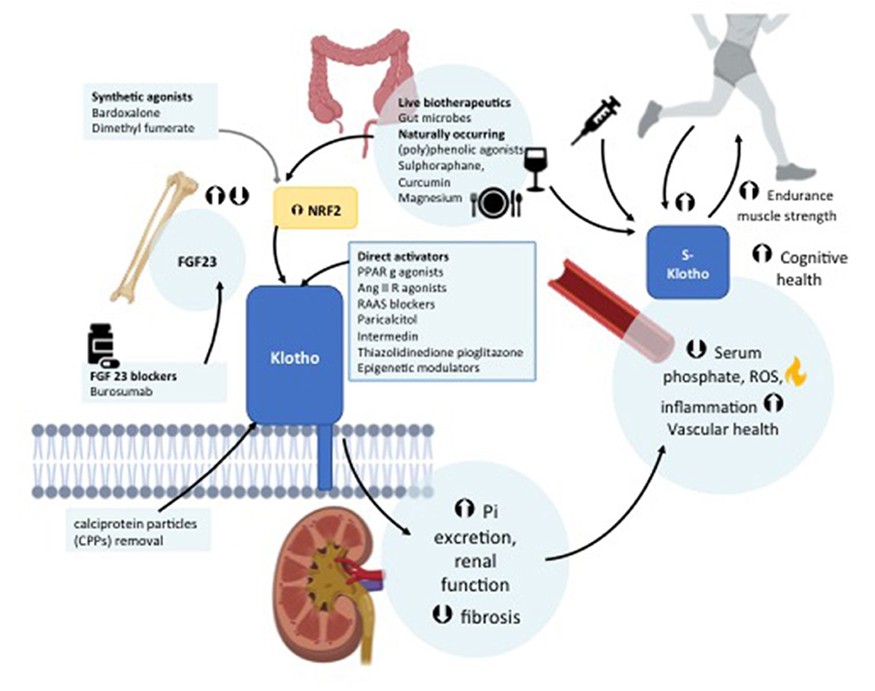
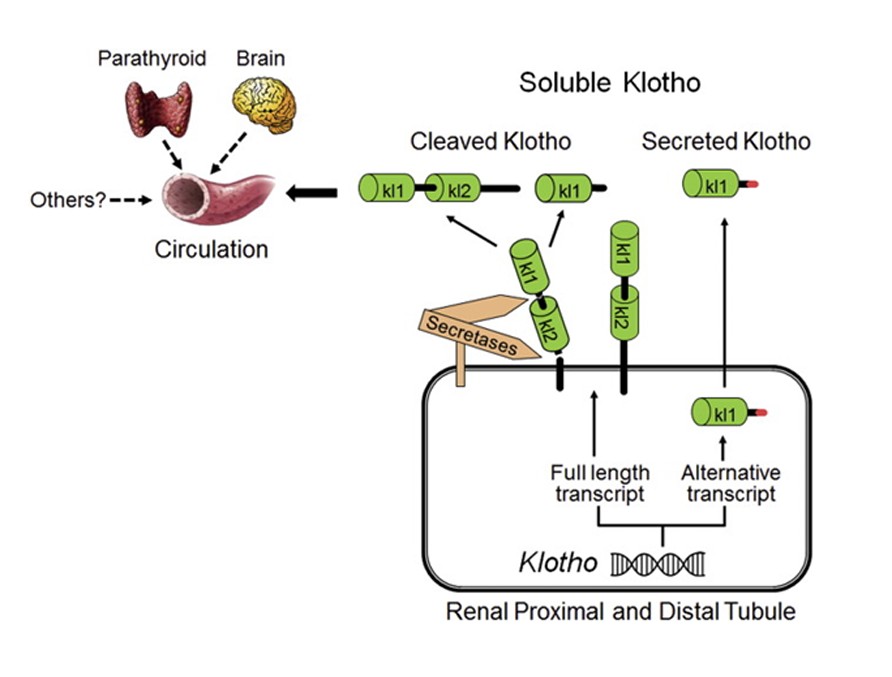
Klotho, a protein named after the Greek goddess of fate, has garnered significant attention in recent years for its role in kidney health and diseases. Klotho exists in both membrane-bound and soluble forms, each playing distinct roles in the body. Predominantly expressed in the kidneys, klotho functions as a co-receptor for fibroblast growth factor 23 (FGF23), regulating phosphate and vitamin D metabolism. Importantly, klotho has emerged as a critical biomarker for kidney diseases and a potential therapeutic target.
Low levels of klotho are closely associated with chronic kidney diseases (CKD). As kidney function declines, the expression of klotho decreases, leading to disturbances in mineral metabolism, vascular calcification, and accelerated aging-like symptoms. Reduced klotho levels not only reflect kidney damage but also exacerbate the progression of CKD by promoting fibrosis, inflammation, and oxidative stress. Thus, klotho deficiency is both a consequence and a contributor to kidney diseases, making it a valuable indicator of renal function.
Conversely, enhanced klotho levels are indicative of better kidney function and overall health. Higher klotho levels have been linked to protective effects against kidney injury, including anti-inflammatory, anti-oxidative, and anti-fibrotic properties. These beneficial effects contribute to the maintenance of renal function and slow the progression of kidney diseases. Therapeutic strategies aimed at increasing klotho levels or mimicking its action are being explored as potential treatments for CKD, highlighting the protein's crucial role in renal health.
In summary, klotho serves as a vital biomarker and regulator of kidney function. Low klotho levels are a clear indication of kidney diseases and its progression, while enhanced klotho levels are associated with better kidney function and protection against renal damage. Understanding and manipulating klotho levels could pave the way for novel therapeutic approaches to combat kidney diseases and improve patient outcomes.

Extensive clinical research has demonstrated the critical role of Klotho in chronic kidney diseases progression and patient outcomes. The following landmark studies highlight the growing evidence for Klotho as both a biomarker and therapeutic target in kidney diseases management.
Am J Nephrol (June 2024) 55 (3): 284–286 (American Journal of Nephrology)
https://pubmed.ncbi.nlm.nih.gov/38198777/Institute Journal of Molecular Science 3rd February 2024
https://www.mdpi.com/1422-0067/25/3/1843Sci Rep 2024 Feb 23;14(1):4477
https://pubmed.ncbi.nlm.nih.gov/38396063/Clinical Kidney Journal, Volume 17, Issue 1, January 2024
https://pubmed.ncbi.nlm.nih.gov/38213484/Scientific Reports 08 March 2024
https://www.nature.com/articles/s41598-024-56377-8Kidney Blood Press Res 2023 Apr 5
https://pubmed.ncbi.nlm.nih.gov/37019097/Antioxidants (2023) 12(2), 239;
https://doi.org/10.3390/antiox12020239Front. Public Health, 06 October 2022
https://pubmed.ncbi.nlm.nih.gov/36276390/Serum soluble Klotho levels were positively associated with eGFR and inversely associated with the prevalence of CKD stage 3 or more. Dose-response relationships were observed between Klotho and the occurrence of CKD stage> 1 and albuminuria.
Am J Nephrol (2024) 55 (3): 273–283.
https://pubmed.ncbi.nlm.nih.gov/38104542/Clin J Am Soc Nephrol. 2021 Jan 7; 16(1): 162–176.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7792642/Front. Endocrinol., 27 August 2020
https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2020.00560/fullKidney Dis (2020) 6 (6): 395–406.
https://doi.org/10.1159/000509369J Nephrol. 2010 Nov-Dec; 23(Suppl 16): S136–S144
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3227531/J Am Soc Nephrol. 2014 Oct; 25(10): 2169–2175.
https://pubmed.ncbi.nlm.nih.gov/24854271/BMC Nephrology volume 19, Article number: 285 (2018)
https://bmcnephrol.biomedcentral.com/articles/10.1186/s12882-018-1094-zBone Volume 100 , July 2017, Pages 41-49
https://www.sciencedirect.com/science/article/abs/pii/S8756328217300170