People worldwide are living longer, with most individuals now expected to live into their sixties and beyond. Every country is witnessing an increase in both the size and proportion of older persons within the population.
By 2030, 1 in 6 people globally will be aged 60 years or over. The share of the population aged 60 and above will rise from 1 billion in 2020 to 1.4 billion. By 2050, the number of people aged 60 years and older is projected to double to 2.1 billion. Furthermore, the population of individuals aged 80 years or older is expected to triple between 2020 and 2050, reaching 426 million.
At the biological level, ageing results from the impact of the accumulation of a wide variety of molecular and cellular damage over time. This leads to a gradual decrease in physical and mental capacity, a growing risk of diseases and ultimately death.

According to a PubMed published study "Is Aging a diseases? A Critical Review Within the Framework of Ageism", WHO currently considers aging as a diseases.
According to another published study “Hallmarks of aging: An expanding universe”, aging is driven by hallmarks fulfilling the following three premises: (1) their age-associated manifestation, (2) the acceleration of aging by experimentally accentuating them, and (3) the opportunity to decelerate, stop, or reverse aging by therapeutic interventions on them
Damage to our DNA.
Changes in gene expression without altering the DNA sequence.
Impaired cellular cleaning processes.
Accumulation of aged, non-dividing cells.
Persistent, low-level inflammation.
Shortening of the protective caps on our chromosomes.
Problems with protein maintenance and balance.
Issues with how cells sense and respond to nutrients.
Depletion of stem cells needed for tissue repair.
Imbalance of gut bacteria.
Decline in the function of cellular energy producers.
Problems with cell-to-cell communication.
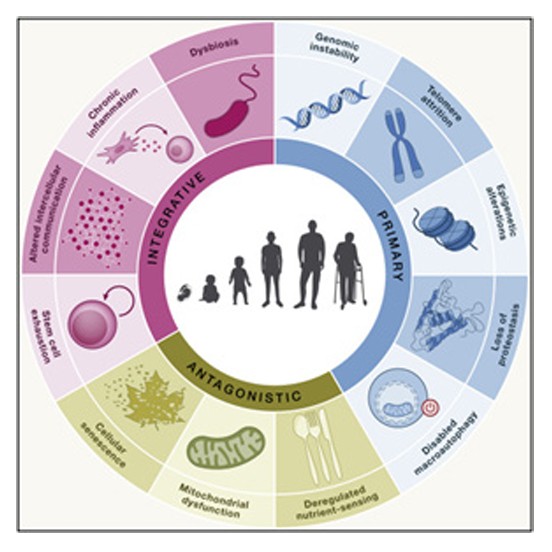
These hallmarks are interconnected among each other, as well as to the recently proposed hallmarks of health, which include organizational features of spatial compartmentalization, maintenance of homeostasis, and adequate responses to stress.
Low Klotho levels are strongly associated with accelerated aging and a shorter lifespan. Individuals with lower levels of Klotho may experience an earlier onset of age-related conditions such as cardiovascular diseases, osteoporosis, and cognitive decline.
The deficiency of Klotho can lead to increased oxidative stress and inflammation, further exacerbating the aging process. Additionally, reduced Klotho levels are linked to impaired kidney function and a higher risk of developing chronic kidney diseases, which is a significant contributor to overall health decline in aging individuals. Maintaining adequate Klotho levels is crucial for promoting longevity and mitigating the adverse effects of aging.
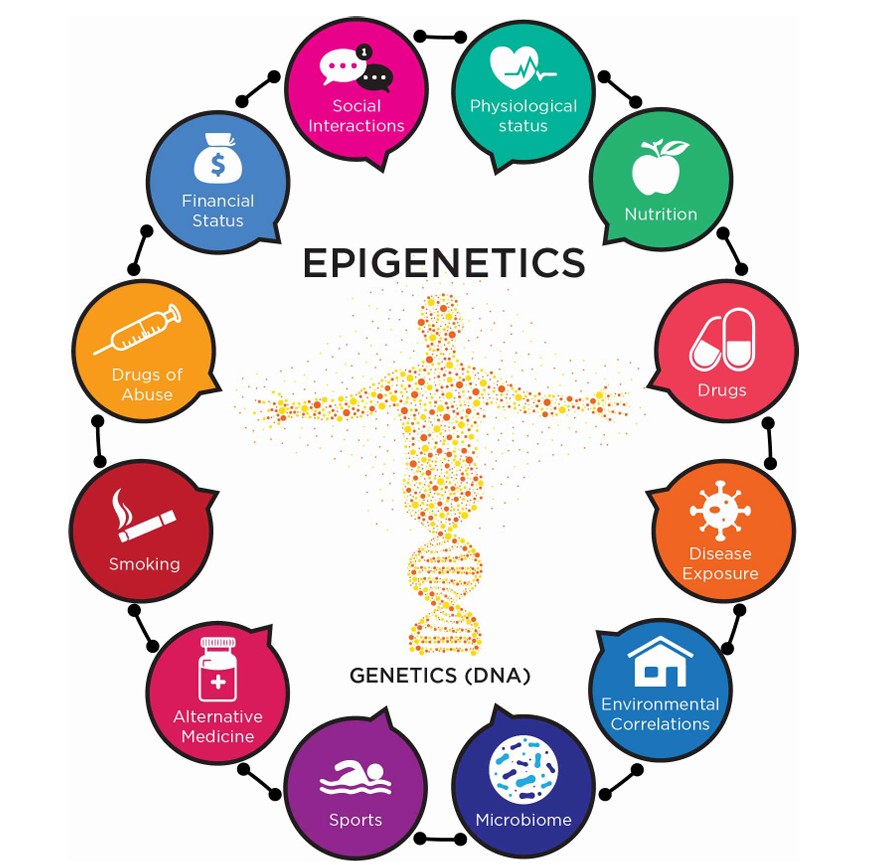
While medical advances have significantly extended life expectancy, the prevalence of chronic diseases and geriatric syndromes continues to rise among the aging population. Despite this, modern medicine often focuses more on treatment than on prevention and education. To address this growing issue, a new field known as lifestyle medicine has emerged - Epigenetics.
Epigenetics refers to how your behaviours and environment can cause changes that affect the way your genes work. It is the study of how cells control gene activity without changing the DNA sequence. "Epi-"means on or above in Greek, and "epigenetic" describes factors beyond the genetic code. Epigenetic changes are modifications to DNA that regulate whether genes are turned on or off.
These changes can affect how cells read genes and are influenced by various factors, including environmental stimuli, lifestyle, and diseases states.
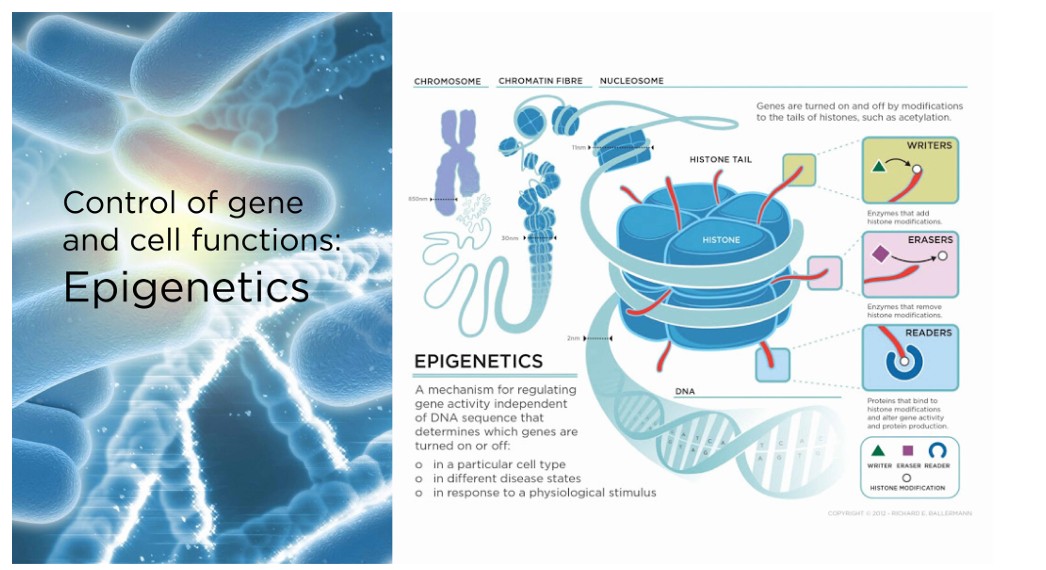
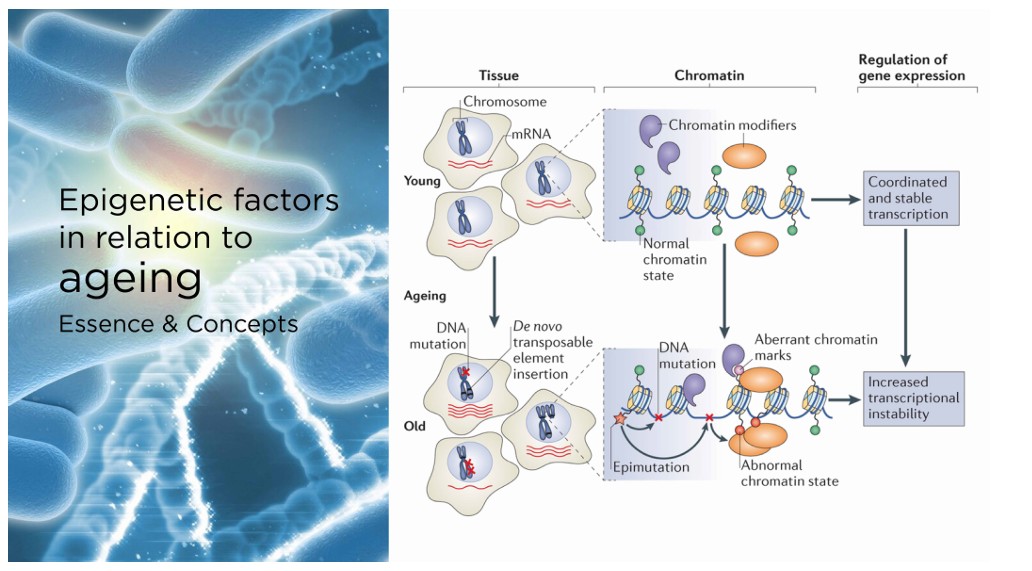
The specific role of the Klotho protein in human health and diseases remains somewhat ambiguous. Below are several factors and complementary approaches that may boost Klotho levels. However, more extensive studies are needed to draw definitive conclusions.
Both humans and mice show increases in Klotho levels after exercise. Notably, a 16-week training program resulted in higher Klotho levels, especially in younger individuals. Research suggests muscle injury and the formation of new muscle cells may promote Klotho secretion.
While calcitriol can increase Klotho, vitamin D supplements do not seem to have the same effect in humans and can even decrease Klotho levels in dialysis patients. Calcitriol is a synthetic derivative of Vitamin D3 that aids in treating metabolic bone diseases and calcium insufficiency.
Rapamycin-treated mice exhibited significantly enhanced autophagy and elevated expression of klotho protein, particularly in the kidney.
Insulin has been shown to increase Klotho, which is a consideration for those on low-carb diets.
These compounds have been linked to increased Klotho levels. Peroxisome proliferator-activated receptor gamma (PPARG) increases insulin sensitivity by enhancing storage of fatty acids in fat cells, enhancing adiponectin release, inducing FGF21, and enhancing nicotinic acid adenine dinucleotide phosphate production.
This can bind to indoxyl sulphate, a toxin that decreases Klotho, potentially preventing its negative effects.
Certain probiotics, such as Acidophilus and L. Lactis, have increased Klotho levels in aging mice.
This supplement has been shown to reverse the decrease in Klotho caused by angiotensin II.
This may help stabilize the process involved in Klotho production.
These medications may have a positive impact on Klotho levels.
These drugs may help stabilize Klotho after its production.
While these factors show potential, definitive evidence is still lacking, and professional medical advice is crucial before making any changes.
Klotho enhances the expression of antioxidative enzymes and reduces the production of reactive oxygen species (ROS). Lower oxidative stress helps protect cells from damage, a key factor in slowing down the aging process.
Klotho influences insulin signalling, which is a critical pathway that impacts metabolism and lifespan. It can increase insulin sensitivity, thereby improving glucose metabolism.
Klotho modulates the insulin-like growth factor 1 (IGF-1) pathway, which is known to affect aging and growth. By reducing IGF-1 signalling, Klotho can contribute to extended lifespan and reduce aging-related diseases risks.
Klotho functions as a co-receptor for FGF23, a hormone involved in phosphate regulation. Proper regulation of phosphate levels is crucial for preventing calcification in tissues and maintaining cellular health, thereby influencing longevity.
Klotho regulates calcium channels and transporters, helping to maintain calcium balance in the body. This balance is essential for vascular and skeletal health, both of which are important in aging populations.

Klotho has been shown to reduce cellular senescence, the state where cells stop dividing and accumulate toxic compounds. This prevention is crucial for maintaining tissue and organ function over time.
Klotho can also inhibit apoptosis or programmed cell death in various cell types, thus preserving cell longevity and functionality.
Klotho downregulates pro-inflammatory cytokines and upregulates anti-inflammatory cytokines. Chronic inflammation is a hallmark of aging, and its reduction is beneficial for extending healthspan and lifespan.
High levels of Klotho have been associated with better cognitive function and protection against neurodegenerative diseases. It supports brain health by reducing neuroinflammation and enhancing neuronal survival.

Klotho helps reduce arterial stiffness and suppresses the formation of atherosclerotic plaques, promoting a healthier cardiovascular system, which is vital for longevity.
In essence, Klotho acts on multiple fronts to mitigate the adverse effects of aging by regulating key metabolic, cellular, and molecular pathways. This multifaceted approach helps to preserve physiological functions, slow down age-related decline, and ultimately extend lifespan.